The bit of advice I struggle with the most is around cooking oil… there so much conflicting information out there and the science can be difficult to understand. Plus it is well established in many cultures to cook with these oils and many people feel that if they are in the supermarkets, then they must be ok. Doing biochemistry revision for my nutrition exam on Saturday has reminded me just how important this issue really is and how misinformed we are.
Many vegetable oils such as canola, sunflower, soy and sesame seed are not only highly processed and very high in omega 6, but is also very prone to oxidative stress and can form free radicals and here is a brief summary of what that means…
Processing
Vegetable oils are often not made from the pure original source material. For example, canola is isn’t normally made form the canola seed, but a hybrid, most often a genetically modified form of rapeseed. So you’re already starting with an unnatural, tampered seed that potentially has toxic chemicals such as pesticides in them. The seeds are often heated to very high temperatures, destroying the nutrient content and oxidising them (more about this below) and also sometimes petroleum or other harmful products can be used to extract the oils. Acids can be used to take away waxy bits and further chemical processing can be used to mask smells and improve the colour. By the time you get the oil it’s gone through so much processing, you really don’t know what it really is any more. It’s certainly no where near cold-pressed!

Cold-pressed is a safe and toxin free way of extracting the oil from a substance and gives you the first extract. This is the pure juice of the substance. For example, extra virgin olive oil. However, this is something to consider carefully when out shopping, are you really buying cold-pressed pure oil? I’ve seen oils like pomace olive oil which is basically the oil extracted from the pulp of the first cold-pressed olives but solvents are used to extract this. We may be unknowingly buying a product that is not pure, and that may not be what we want.
Oxidative Stress
And now for a bit of science… Vegetable ois are what we call polyunsaturated fats (a type of fat). These fats are made up of groups of carbon and hydrogen molecules (CH2 groups) with at least 2 double bonds which means they are missing at least 2 hydrogen molecules. This makes the bonds very vulnerable to breakage leaving the CH2 groups exposed to other atoms and molecules coming and stealing the electrons on their outer shell (free radicals). They themselves also become free radicals and steal other atoms and molecule’s electors, continuing the chain of reaction. These free radicals may be stealing electrons from atoms that we need in our body and so they will not be effective. Furthermore, these ineffective atoms could become dangerous in their own right. Heating these oils when cooking makes this process more overwhelming and can cause damage to our cells in the long term.
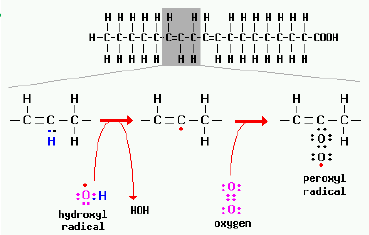
Omega 3s & 6s
We should be eating about the same amount of omega 3s to omega 6s, so that we have the right balance of anti-inflammatory EFAs (essential fatty acids) rather than some anti-inflammatory omega 3 EFAs and quite a lot of omega 6 EFA which become pro-inflammatory when eaten in excess. However with polyunsaturated fats hidden in processed foods, the typical western diet sees us consuming much more than this, around a ration of 1:15 Omega 3s:6s and for some people, even more. Whilst omega 6 fats (which are polyunsaturated fats) have their beneficial properties, when consumed in excess it becomes a problem causing chronic inflammation. Chronic inflammation is what leads to more serious health problems down the line.
Omega 3 oils can be found in flaxseeds, avocado and fish. These oils are not generally suggested for cooking for a variety of reasons, the most important one being that they are too precious and should be consumed in as pure a form as possible to ensure the best nutritional value is retained. Cooking and eating oily fish high in omega 3 is fine, especially with the bone left on as this also has other important nutrients.
So what should I use?
Even the finest first, cold-presses oils may not be appropriate for some methods of cooking. We need to be aware of cooking methods and which oils are appropriate for what. It all comes down to how stable the oil is at different temperatures. When oils are overheated they essentially burn and go rancid, and often you can’t smell or taste a difference. If you see smoke coming from a pan with pool in it, even if it is very light or faint, then it’s already burnt and rancid. Throw it away!
Olive oil is fine up to about 270 degrees Celsius so using it for light sautéing and simmering is fine. Obviously, using it in combination with raw foods such as in salads is perfect.

Vegetable oils are really unstable as we’ve discussed earlier and have a very low burning temperature so best to keep them for raw food combinations too.
Saturated fats such as coconut, palm, butter, animal fats (like goose fat) and ghee have all their carbon bonds filled with hydrogen bonds and no double bonds. This means they are much more stable when heated and can withstand higher temperatures. These oils should be used for cooking methods that require higher temperatures such as roasting. The great thing about saturated fats is that they are good for us despite what bad press they get, along as you are eating moderate amounts, in combination with a balanced diet and not burning them!

It’s not easy to sift through all the information out there and I hope I’ve been able to make it a little easier. Below are some interesting articles and I’ve thrown in an interesting medical journal in there as well! Enjoy the Youtube video which also explains things in simple terms.
Peace
[youtube https://www.youtube.com/watch?v=E3JkXdjQJJg]
References
Taylor H & Cotton D eds (2014) Biochemistry: The study of chemical compounds and reactions in living systems, College of Naturopathic Medicine
Mercola J, The science is practically screaming… don’t make this trendy fat mistake
The ugly truth about vegetable oils (and why they should be avoided)



